Home » Warfarin: The original anticoagulant

Learning outcomes:
At the end of this article you will be able to:
Warfarin is a coumarin oral anticoagulant and prior to 2012, was the mainstay for the treatment and prevention of VTE, and other indications such as heart valves that require anticoagulation, until the introduction and reimbursement of the Direct Acting Oral Anticoagulants (DOAC) apixaban, dabigatran, edoxaban, and rivaroxaban.
Warfarin is a vitamin K antagonist and exerts its action on the clotting cascade by inhibiting vitamin K dependant clotting factors; II, VII IX and X, resulting in decreases in these clotting factors. By depleting these clotting factors, blood is less able to clot. Whereas warfarin lowers the production of a number of clotting factors, DOACs directly inhibit thrombin (dabigatran) or Factor Xa (apixaban, rivaroxaban, edoxaban). As a result, warfarin requires ongoing monitoring and possible dose adjustments because of its broader effect on clotting, compared to DOACs and are given as fixed doses.
Warfarin is readily absorbed, but has a delayed onset of action, at about 48 hours (24 – 72 hours). It is given as a once-a-day dose. Steady state levels are not reached for up to eight days and antithrombotic action is not typically present until day five. Hence, when starting patients on warfarin, loading doses are often used to achieve therapeutic levels.
Warfarin is almost entirely metabolised in the liver, primarily via the Cytochrome P450 enzymes (CYP 1A2, CYP2C9, CYP3A4). Numerous drug interactions have been documented with warfarin, and inhibition and induction of the Cytochrome P450 enzymes are frequent mechanisms of interaction. The metabolites of warfarin are renally excreted.
Genetic polymorphisms significantly affect individual responses to warfarin, further contributing to unpredictability and challenges in managing anticoagulation.
The effectiveness of warfarin is monitored by a blood test called prothrombin time (PT), standardised as the International Normalized Ratio (INR ). This measures how long it takes for the blood to clot. The INR of a healthy patient not on anticoagulation therapy is approximately 1.0. Therefore, a patient with an INR of 2.0 or 3.0 requires two or three times longer for their blood to clot. The therapeutic INR goal for patients on warfarin therapy depends on the indication, as outlined later in this article.
After discontinuation of warfarin dose, INR typically takes five days to return to normal.
Warfarin is indicated for use in adults for the management and prevention of venous thromboembolism (VTE), this includes:
Initiating therapy is a fine balance between ensuring a therapeutic INR is reached in a timely manner to prevent thrombosis, while avoiding excessive anticoagulation and haemorrhage. A number of baseline tests should be carried out prior to starting warfarin:
A variety of warfarin loading protocols are in use. The BNF suggests a starting dose of 5-10mg on day one, with subsequent doses based on INR.
Other resources suggest a lower induction dose (2mg once daily), to be given over three to four weeks for patients who don’t require rapid anticoagulation with weekly INR checks and dose adjustments as required.
The dose should be taken once a day at a fixed time, preferably 18.00 hours, or another regular time if more convenient, to aid compliance. Patients are mostly seen by their prescriber during the day, therefore a late afternoon or evening dose enables their prescriber to ask the patient to miss a dose when required, for example, if INR is too high.
Table 1: Indication for warfarin, target INR and duration of therapy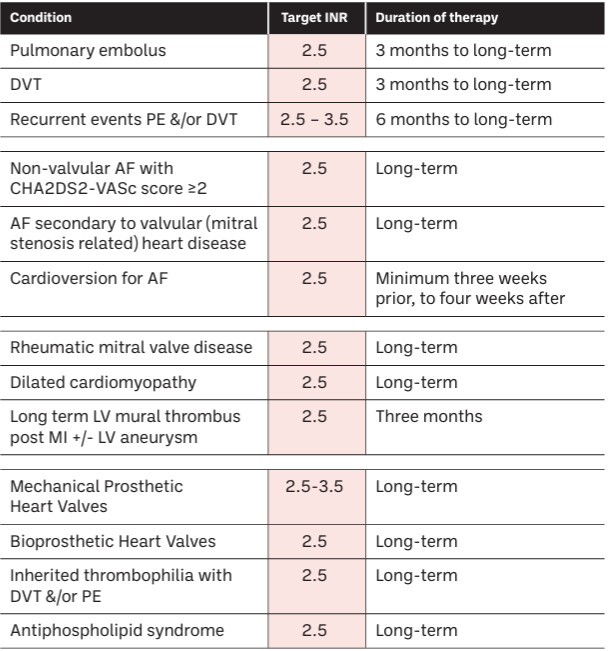
Anticoagulant duration for VTE typically starts at three to six months but extends indefinitely for high-risk cases like cancer-related clots or second unprovoked events, balancing clot recurrence risk against bleeding risk, with factors like patient gender, event type (PE vs. DVT), and bleeding risk influencing the decision. A first, provoked VTE (due to a temporary trigger like surgery or COC), usually stops at three months, while unprovoked events often lead to longer therapy, often indefinite. It should also be noted that when used for the prophylaxis of VTE in patients with atrial fibrillation and prosthetic valves, warfarin will be prescribed on a long-term, possibly indefinite duration.
The BNF advises that INR should be checked daily or on alternate days in the early days of treatment, then at longer intervals, depending on response to treatment, and then up to every 12 weeks.
Should there be any changes to the patient’s clinical condition, this will necessitate more frequent testing; changes may include intercurrent illness, liver disease or altered liver function, or changes to patient’s other medicines (addition, discontinuation or dose changes).
It must also be noted that dose adjustments can take 48 hours to take effect so a meaningful INR can only be obtained after three to four days, following warfarin dose changes.
For some patient groups, they will require more frequent INR checks: these groups include those at risk of over-anticoagulation (for example, severe, uncontrolled hypertension, liver disease, renal failure); or if there is an increased risk of bleeding (for example, age 65 years or over, highly variable INRs, history of GI bleeding, cerebrovascular disease, serious heart disease, excess alcohol intake, risk of falling etc.).
Bleeding is the most common adverse effect of warfarin and may occur at therapeutic INR. Gastrointestinal and cerebral bleeds are most commonly seen.
Skin reactions are a common side effect: purpura (small blood spots) and ecchymoses (bruising and skin discolouration) are common in over-anticoagulated patients. Skin necrosis, vasculitis and pruritic skin rashes have been reported, including macular, papular, vesicular and urticarial lesions. Patients are advised to contact their doctor if they develop a painful rash while on warfarin.
Skin necrosis is a rare but serious side effect of warfarin. It occurs mainly in obese, female patients, usually within three to 10 days of starting therapy. Initially, the lesions consist of painful, indurated, reddened areas, which progress through a stage of blood-filled blisters into well-demarcated blackened necrotic patches. Areas of skin with underlying fatty tissue, such as breasts, flanks and buttocks are most often affected.
Purple toes syndrome is a rare complication of warfarin therapy. Typically, the syndrome presents three to eight weeks after initiation of warfarin therapy as a sometimes painful, blue-tinged discoloration of the plantar aspects and sides of the toes.
Calciphylaxis is another rare syndrome of vascular calcification with cutaneous necrosis, associated with high mortality. Rare cases of calciphylaxis have been reported in patients taking warfarin.
Rare or very rare side effects include alopecia, nausea and vomiting.
Patients who present with any of the above should be referred to their GP as a matter of urgency.
Because warfarin has a narrow therapeutic window, drug interactions can have a significant impact on anticoagulation control. Induction and inhibition of the cytochrome P450 enzyme subtypes (CYP 1A2, CYP2C9, CYP3A4) is a frequent mechanism of interaction, resulting in low and high INR, respectively.
Drugs which are liver enzyme inhibitors interact with warfarin and increase the INR, and drugs which are liver enzyme inducers interact with warfarin and decrease the INR. There are other drugs which have other mechanisms of interaction, which further increase the risk of bleeding in patients on warfarin, often synergistically, where the sum of the risk is greater than with either drug alone (see Table 2). Patients should not be on both a DAOC and warfarin and any prescriptions for the use of the two medicines together should be queried.
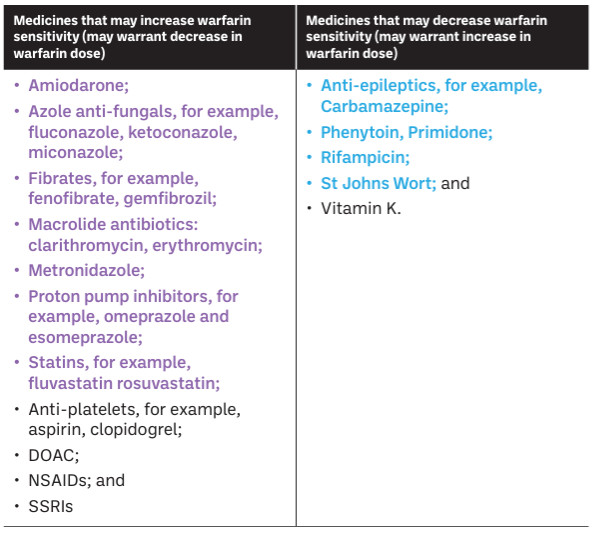 Table 2: Drug interactions (use colours as below)
Table 2: Drug interactions (use colours as below)
Note:
Where a patient already on warfarin therapy is commenced on a long-term enzyme inducer or inhibitor, the patients INR may be monitored more closely initially and the warfarin dose can be adjusted accordingly to ensure a therapeutic INR is achieved and maintained.
Changes to lifestyle and diet food may also impact on the INR.
Table 3: Food and conditions that may affect warfarin sensitivity
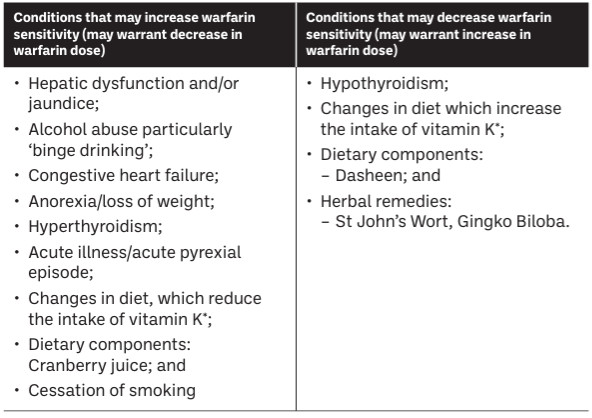
Patients on warfarin therapy should be advised to check with their pharmacist when considering the use of a multivitamin supplement, fish oils or omega 3 fatty acids, as many may contain vitamin K.
The lists above are not exhaustive, and it is good practice to check for potential drug interactions, when any new drug is prescribed for a patient taking warfarin, or the patient flags any changes in diet and lifestyle, additions of dietary supplements, oral nutritional supplements or OTC products.
Warfarin is a known teratogen and should be avoided in pregnancy. Women of childbearing age on warfarin should be warned of teratogenicity and advised on the use of adequate contraception or to discuss choice of anti-coagulant if planning a pregnancy.
Warfarin is not seen in significant quantities in breastmilk and so appears safe for use in breastfeeding. It is advised to monitor closely for any signs of bleeding in the nursing mother or baby.
The IPU Professional Academy Autumn 2025 series included the topic ‘Medicines use in Pregnancy’.

Sinéad McCool MPSI

Professional Services, IPU
Highlighted Articles