Home » The fight against medicine shortages: EC publishes Critical Medicines Act

The Critical Medicines Alliance (CMA) was established in January 2024 by the European Commission to help tackle medicine shortages, which are affecting all EU countries. It aims to bring together stakeholders from EU Member States, key industries, civil society bodies, and the scientific community, to identify key areas and priorities for action. An overarching objective of the CMA is the need for enhanced coordination between stakeholders, Member States and the European Commission. The Commission says the CMA is a key tool in the fight against the ongoing problem of medicine shortages.
The CMA objectives are to:
An article on the launch of the Critical Medicines Alliance was published in the July/August 2024 issue of the IPU Review, available to IPU members at ipu.ie/ipu-review > Archive Articles.
Medicine shortages have been a key concern in Ireland and across EU Member States for the past number of years. The Critical Medicines Alliance was established in January 2024 as a means to enhance coordination between Member States as the European Commission declared that “no single entity can solve these challenges alone”. In European Commission President Ursula von der Lyden’s statement on Political Guidelines for the next European Commission 2024-2029, she said, “We must make our economy more resilient and less dependent. This is notably important in the health and pharmaceutical sector.” To remedy the “severe shortages of medical devices and medicines” the EU is facing, she said the Commission would propose a Critical Medicines Act “to reduce dependencies relating to critical medicines and ingredients, particularly for products where there are only a few supplying manufacturers or countries”.
Image 1: Root causes of medicine shortages for 2022 and 2023 in EU/EEA countries

Source: European Commission
The Strategic Report of the Critical Medicines Alliance was published on 28 February. The Report outlines key findings and recommendations to enhance the security and resilience of the EU’s critical medicines supply chains.
It highlights the critical vulnerabilities within Europe’s pharmaceutical supply chains, and proposes substantial investment in strategic projects. It also addresses procurement policies, while advocating for a harmonised and balanced framework of stockpiling requirements to ensure the security of supply. It also recommends strengthening partnerships with third countries to bolster supply resilience.
The Critical Medicines Act was proposed by the European Commission on 11 March, saying it will “improve the availability, supply and production of critical medicines within the EU”. The Act includes the following key features:
Image 2: Key elements of the Critical Medicines Act
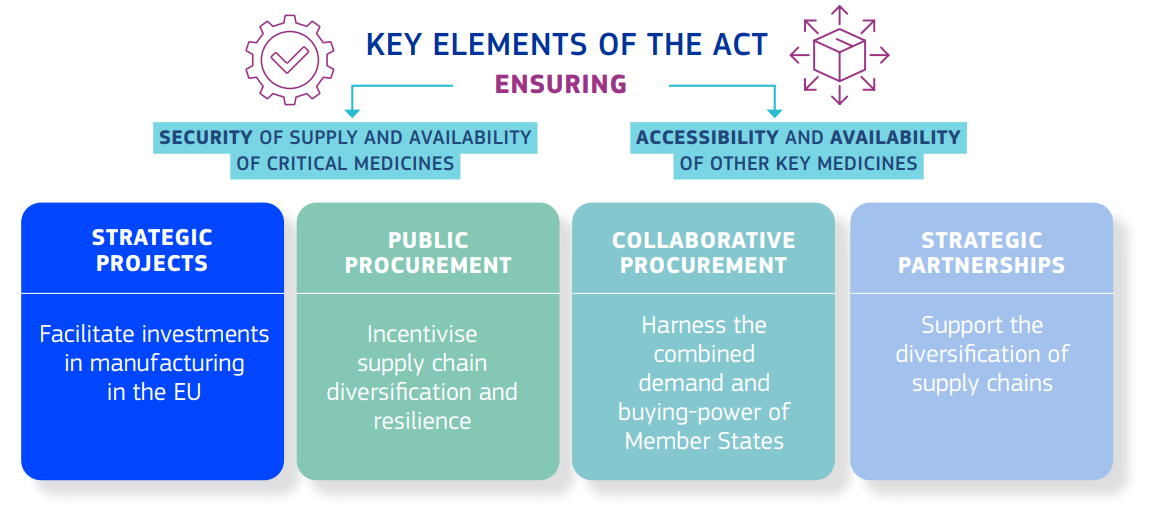
Source: European Commission
The Act will cover critical medicines, for which limited or no alternatives exist and shortages of supply may result in serious harm to patients; critical medicines for which vulnerabilities have been identified in their supply chain, notably high dependencies on a single or limited number of third countries; and certain other medicines of common interest, for example for rare diseases, which may not yet be accessible in certain markets.
The Act will facilitate investment in manufacturing in the EU, by supporting ‘Strategic Projects’. Strategic Projects are industrial projects for critical medicines, which “create, increase or modernise manufacturing capacity in the Union”. Such projects will have easier access to funding, and fast-tracked administrative, regulatory and scientific support. In return, if companies have benefitted from financial support for a Strategic Project, they will be obliged to prioritise EU supply.
The Act aims to incentivise supply chain diversification and resilience and will provide for this by introducing new procurement guidelines for Member States and procurers. This will mean that procurers will have to apply procurement requirements other than price in their public procurement procedures for critical medicines. Procurers will also have to favour EU production for specific critical medicines with high dependencies and may do the same for other medicinal products of common interest, when justified.
The Act allows EU countries to request European Commission support to use different collaborative procurement tools. This will allow for enhanced purchasing power to create economies of scale and reduce access disparities.
The EU will explore partnerships with like-minded countries/regions to diversify the supply chain and reduce dependencies on single suppliers.
The Department of Health, along with the Department of Enterprise, Trade and Employment and the HPRA represented Ireland and participated in various Working Groups of the Critical Medicines Alliance, and it welcomed the CMA’s Strategic Report. Speaking to the IPU Review, the Department says it, “supports the overall objectives within the report of access to Critical Medicines for EU citizens and ensuring European open strategic autonomy.”
The Department says the Strategic Report and the work of the Critical Medicines Alliance, is represented in the European Commission’s proposal for a Critical Medicines Act; “Ireland was one of many Member States who called for such a legislative initiative by giving support to Belgium’s 2023 non-paper Improving the security of medicines supply in Europe. The Critical Medicines Act provides a further opportunity to progress key objectives for patients, national health services and the pharmaceutical industry, in the context of supporting the supply of medicines.”
The Department of Health says it will, “work with key stakeholders to inform and support Ireland’s input into the consideration of the Act and looks forward to beginning work on this legislative initiative via the Council Working Party on Pharmaceuticals and Medical Devices”.
The Department concluded by saying it “is aware of the stress that shortages of medicinal products can cause for patients and healthcare professionals and therefore looks forward to continued engagement in European initiatives aimed at tackling the underlying causes of recurrent medicine shortages”.
The Pharmaceutical Group of the European Union (PGEU) is the voice of community pharmacy in Europe, and they also welcomed the Act, calling it a “timely proposal . . . aimed at addressing the urgent and complex issue of medicines shortages”. The representative body said that by focusing on the root causes of shortages, “this regulation offers a pathway to ensuring patients can access the critical medicines they need”.
The need for “autonomy in the current geopolitical context” was also referenced, as the PGEU asserts that the Critical Medicines Act can do this “by introducing incentives for strategic projects and other forms of financial support, aimed at enhancing European production capacities and diversifying supply”.
However, the PGEU emphasises that reducing dependence on third countries must go hand in hand with carefully balancing affordability and security of supply; “Incentives reinforcing European production should be cost-effective, targeted, and sustainable in the long term. Any public investment to strengthen the manufacturing of critical medicines must be linked to clear and enforceable industry commitments that guarantee uninterrupted supply for European patients. Additionally, authorities must be further empowered to oversee supply obligations, enforcing compliance where necessary.”
The PGEU also supports the Commission in making use of the recently announced, EU defense spending package, to finance the provisions of the Critical Medicines Act.
They did however, caution against duplication of stakeholder groups, as the proposal introduces a new Critical Medicines Coordination Group; “While PGEU acknowledges the intent to reinforce coordination at EU-level, there is concern about potential overlap with the existing Medicines Shortages Steering Group (MSSG). Ensuring alignment between these bodies and avoiding duplication is vital to a swift and effective implementation of the Act.”
Emphasising that community pharmacists are battling medicine shortages everyday, the PGEU said, “European community pharmacists stand ready to support the Critical Medicines Act and will continue contributing daily to effective solutions to combat shortages. A successful resolution of this systemic challenge is crucial to preserving the continuity and quality of care that European patients expect.”
The European Patients’ Forum (EPF) is an umbrella organisation of patient organisations across Europe and across disease areas. The EPF welcomed the European Commission’s proposal for the Critical Medicines Act as, “an important step toward improving medicine security, availability as well as accessibility across the EU for all patients”. However, the EPF says the Act also “leaves serious gaps that must be addressed to ensure its effectiveness”: “For the Act to succeed, it must move beyond broad ambitions and commit to tangible actions.”
The EPF identifies medicine shortages as a major public health problem “that can seriously harm patients’ health and safety, quality of care, professionals’ ability to deliver care, and health system functioning”. In the terms of the proposed Act, the EPF says ensuring security of supply, availability and accessibility of critical medicines and medicinal products is vitally important, as is the inclusion of public procurement based on criteria other than price. The EPF also welcomes the commitment to evaluate the Act’s impact after five years with a structured mechanism to assess progress and make necessary improvements.
However, it identifies some issues, including finance; “We are very concerned about how these initiatives will be financed in the short and longer term . . . while the focus is currently on defence and competitiveness, strong and resilient healthcare systems should be an integral part of a comprehensive security strategy.”
The EPF also calls for a strong EU coordination mechanism for stockpiling, “to avoid duplication, ensure consistency, and facilitate reallocation of stocks between EU Member States”.
IPHA, which represents the international research-based pharmaceutical industry in Ireland, also welcomed the Critical Medicines Act: “Ensuring timely access to critical medicines is a shared priority of the European Commission and the biopharmaceutical industry. To achieve this, the policy solutions will need to be targeted, evidence-based, and aligned with international commitments to avoid unintended consequences for supply sustainability and patient access.”
IPHA said its members “remain committed to reducing disparities in access to medicines across Europe, by placing their products on the market as soon as possible”. However, the group also affirms that the Act, “needs to go further and provide an effective response to fragmented national contingency stock requirements that disrupt the functionality of the single market. For example, this could be achieved by making use of existing data systems, such as the European Medicines Verification System (which the Irish Medicines Verification Organisation operates here), to avoid the duplication of reporting requirements. This system provides timely information on the number of packs for all prescription products being uploaded by manufacturers into the European Medicines Verification System (EMVS) . Leveraging this data would reduce the burden on manufacturers and streamline the vulnerability assessment exercise.”
The Critical Medicines Act aims to complement regulatory measures already proposed, especially the reform of EU pharmaceutical legislation. In 2023, the Commission proposed a new Directive and a new Regulation, which revise and replace the existing general pharmaceutical legislation. The Commission says, “The proposed Critical Medicines Act complements the measures already put forward. It presents industrial policy solutions that can work hand-in-hand with the regulatory measures in the proposed pharma reform, to address vulnerabilities in the supply chain of critical medicines and mitigate the risk of shortages. As such, the Critical Medicines Act is part of our work to complete the European Health Union and is fully in line with goals of the Pharmaceutical Strategy for Europe.”
Image 3: European Commission’s Framework to ensure availability of medicines for EU patients

Source: European Commission
The Critical Medicines Act will now proceed through the legislative path in the European Parliament. Further information and updates will provided in the IPU Review as it journeys through Parliament. For further information see health.ec.europa.eu > Medicinal products > Legal framework > Critical Medicines Act.
In May 2023, 23 EU Member States proposed to launch a Critical Medicines Act to diversify the pharmaceutical supply chains. This call was reiterated by the European Council in June 2023 and reinforced in October 2023 (Granada Declaration). In response, the Commission adopted on 24 October 2023 a communication presenting actions to better prevent critical medicine shortages in the EU, published in December 2023 a first version of a Union list of critical medicines and established in 2024 a Critical Medicines Alliance, bringing together Member States, civil society and patients, the scientific community and the pharmaceutical industry. Additionally, the European Economic and Social Committee issued in December 2023 an exploratory opinion ‘Securing Europe’s medicine supply: envisioning a Critical Medicines Act’.
The proposal of a Critical Medicines Act, complementing the proposed revision of the rules on pharmaceuticals, was mentioned by the President of the Commission Ursula von der Leyen in her July 2024 political guidelines. On 6 November 2024, during his confirmation hearing, Olivér Várhelyi, Commissioner for Health and Animal Welfare, committed to tackle the endemic risks of shortages of critical medicines through a Critical Medicines Act, to be proposed by the Commission within the first 100 days of its new mandate. This was confirmed in the Commission’s Work Programme for 2025 published on 11 February 2025. The regulation was then published by the Commission on 11 March 2025.
Siobhán Kane

Editorial Manager, IPU
Highlighted Articles