Learning outcomes
At the end of this article, you will be able to;
- List some common reference sources used for dosing in paediatric oncology;
- Describe the process involved in supplying an unlicensed or manipulated formulation from a paediatric oncology hospital prescription;
- Discuss the most common supportive care medications in children with cancer, including medicines to help prevent infection, manage symptoms, and support recovery during treatment;
- List the key counselling points for oral anticancer agents which are to be administered by families/carers in the home; and
- Outline the safe handling requirements of cytotoxic medicines and the resources available to assist with educating families on protective measures, safe storage, disposal of medicines and bodily fluids, and procedures in case of spills.
In the February issue of the IPU Review, we introduced the Paediatric Cancer Services in Children’s Health Ireland, outlining the structure of the National Children’s Cancer Service (NCCS), the differences between paediatric and adult cancer, and the role of pharmacists within the multidisciplinary team. Based on that overview, this CPD article focuses on the practical aspects of managing paediatric cancer prescriptions in community pharmacy.
Practical considerations in paediatric cancer pharmacy
As an increasing number of children receive oral chemotherapy and supportive medicines at home, community pharmacists play a key role in ensuring the safe and effective use of these treatments.
Community drug schemes
Children diagnosed with acute leukaemia are eligible for the Long-Term Illness scheme, while children diagnosed with cancer within the last five years qualify for a medical card. At the time of diagnosis, hospital social workers support families by initiating applications for the relevant scheme to ensure timely access to medicines. For unlicensed medicines or those not covered by the LTI or GMS, community pharmacists may need to request approval for reimbursement through the Hardship Scheme.
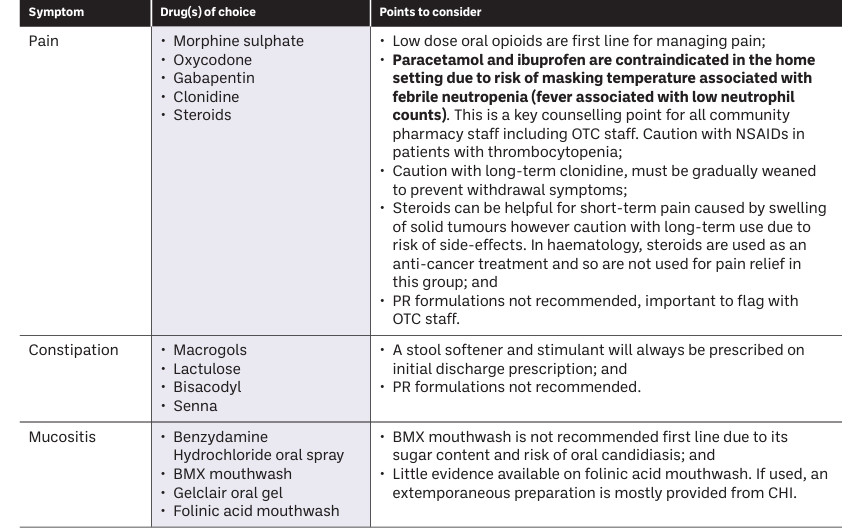
Table 1: Common symptoms and considerations on their management
Paediatric dosing
Paediatric dosing can be challenging, as reliable dosing information is often limited or not readily available. While the British National Formulary (BNF) for Children remains the primary reference for paediatric dosing and indications, prescribing within Children’s Health Ireland is guided by the CHI Formulary. This is developed and maintained by the Pharmacy department and accessible via Clinibee platform.
In paediatric oncology, dosing is guided by chemotherapy protocols, clinical trial requirements, or evidence from the medical literature, and may therefore be challenging to source. To ensure safe and appropriate supply, community pharmacists are encouraged to liaise with the hospital pharmacist to clarify dosing and obtain reference sources used.
Formulation
Many anticancer medicines are developed for adults and so are not available in child-appropriate formulations, such as liquids, dispersible tablets, or low-dose preparations.
While some medicines can be safely crushed or capsules opened, many oral anticancer agents cannot be manipulated, as this can alter bioavailability, reduce efficacy, increase toxicity, or expose carers and healthcare professionals to cytotoxic agents. The British Oncology Pharmacy Association (BOPA) website (see bopa.org.uk), provides a helpful resource outlining oral anticancer medicines that may be safely dissolved or dispersed in water for patients, including children, who are unable to swallow tablets.
In community pharmacy, formulation challenges may be more complex due to challenges in sourcing unlicensed specials, which may have longer lead-times, stock shortages, shorter shelf-life, and may lack consistent stability information. Delays or uncertainty in sourcing these medicines can impact adherence and treatment continuity.
When dispensing unlicensed or manipulated formulations, community pharmacists should:
- Confirm the product has been clinically verified by the hospital haematology–oncology pharmacist;
- Follow specialist guidance regarding storage, stability, and shelf-life;
- Clearly counsel families on safe administration, handling, and storage;
- Avoid crushing or opening tablets or capsules unless explicitly recommended; and
- Liaise promptly with hospital pharmacy if there are concerns regarding formulation suitability, excipients, supply, or stability.
Supportive care medicines
Antifungal prophylaxis
Antifungal prophylaxis is routinely prescribed for high-risk patients. These include children with Aute Myeloid Leukaemia (AML), those with relapsed or progressive disease, infants with Acute Lymphoblastic Leukaemia (ALL), and Bone Marrow Transplant (BMT) patients.
Oral agents available include the ‘azole’ antifungals, for example posaconazole, voriconazole, or itraconazole. Due to their significant interaction profile, including interactions with many chemotherapy agents, a comprehensive medication interaction review is required before starting any azole antifungal.
The formulation prescribed is also an important consideration as azole bioavailability changes between liquid and tablet forms. To ensure efficacy and avoid toxicity, parents should be counselled on the importance of continuing with same formulation and the dietary considerations appropriate for their formulation (for example, posaconazole suspension must be given with high-fat food).
All azole antifungals require close therapeutic drug monitoring (TDM). Sub-therapeutic levels may be caused by gastrointestinal absorption issues, poor adherence to dosing or food requirements, drug interactions, or proton pump inhibitor use. These factors should be reviewed and addressed before dose escalation. Families should be counselled on the purpose of TDM and advised to withhold the morning dose on sampling days.
Antiviral prophylaxis
Post-haematopoietic stem cell transplant (HSCT) and selected high-risk patients require anti-viral prophylaxis, most commonly with valaciclovir or aciclovir. Valaciclovir is the first-line option at CHI as compliance is improved with twice-daily dosing. For patients unable to swallow tablets, the tablets may be crushed and dispersed in water.
Aciclovir liquid may be used when tablet crushing is not feasible, but four-times-daily dosing may be a challenge for adherence.
Important counselling points for antivirals include:
- Ensure child is adequately hydrated; and
- Instructions on crushing if patient is taking valaciclovir.
GCSF
Granulocyte Colony-Stimulating Factor (G-CSF) is a recombinant human growth factor that stimulates white blood cell production, increasing circulating neutrophils. This increase in neutrophils helps reduce the duration of neutropenia and the risk of neutropenic complications, including febrile neutropenia, in children receiving chemotherapy.
In paediatric practice, G-CSF can be given either subcutaneously (SC) or by intravenous infusion (IV), and administration may take place in hospital or at home. The dose, duration, and route depend on the patient’s body weight, the indication, and how the patient responds. G-CSF is available through the High-Tech scheme in short-acting or long-acting (pegylated) formulations. Pegylated G-CSF has a longer half-life and is typically given as a single injection per chemotherapy cycle.
Anti-emetics
Chemotherapy-induced nausea and vomiting (CINV) remains one of the most common adverse effects of cancer treatment and requires proactive management. CINV is classified as acute, delayed or anticipatory, with antiemetics recommended on a round-the-clock basis during chemotherapy administration and for at least 24 hours afterwards. First-line prevention in the acute phase commonly involves 5-HT3 receptor antagonists such as ondansetron or granisetron. Neurokinin-1 receptor antagonists (for example, aprepitant) significantly improve control in highly emetogenic chemotherapy agents when combined with a 5-HT3 antagonist and a corticosteroid, but pharmacists should be mindful of important CYP3A4-mediated drug interactions. Dexamethasone is an effective antiemetic but is contraindicated in several clinical scenarios, including CNS tumours, certain haematological malignancies, and immunotherapy. Second-line agents such as levomepromazine, lorazepam or olanzapine may be useful for refractory symptoms but carry risks of sedation, hypotension, QT prolongation and drug interactions.
PCP and anti-microbial prophylaxis
Children receiving long-term chemotherapy may be at increased risk of developing an uncommon but serious pneumonia known as Pneumocystis pneumonia (PCP). Low dose co-trimoxazole is routinely recommended two or three days per week throughout the course of treatment to reduce the risk of PCP developing.
Some high-risk leukaemia patients, such as infants or those with Down Syndrome, may also require anti-microbial prophylaxis with ciprofloxacin. A typical dose range of 7.5-10mg/kg BD is used. The decision to initiate long-term therapy on quinolones is taken following a benefit-risk assessment of the patient and with close monitoring and counselling on the risk of quinolone-associated toxicity.
Some examples of oral anticancer meds
TKI Imatinib
Tyrosine kinase inhibitors (TKIs) are a newer group of targeted drugs used to treat specific cancers. Tyrosine kinases help to send growth signals in cells. Therefore, inhibiting these signals stops the cell from growing and dividing. Imatinib is a TKI used for Ph+ Acute Lymphoblastic Leukaemia (ALL) and Chronic Myeloid Leukaemia (CML).
Counselling points for Imatinib:
- Swallow the tablet whole, do not crush;
- Counselling on photosensitivity;
- Side-effects are often reversible; and
- Interaction check.
Dabrafenib and Trametinib
Dabrafenib and trametinib are targeted oral therapies commonly used in combination for children with low-grade glioma with BRAF V600 mutations.
Dabrafenib is taken twice daily, 12 hours apart, while trametinib is taken once daily at the same time as either the morning or evening dose of dabrafenib. Both dabrafenib and trametinib should be taken on an empty stomach to ensure optimal absorption. Patients should avoid eating for two hours before taking the medicines and for one hour afterwards. While receiving this treatment, grapefruit and Seville oranges must be avoided in all forms, including fresh fruit, juices and supplements, as they can increase dabrafenib levels in the body and raise the risk of side effects.
Dexamethasone
Dexamethasone is widely used as an anticancer treatment in haematology and as an anti-emetic in oncology.
It should be taken with or after food, and a proton pump inhibitor may be required. Although prescribed twice daily to improve tolerability, doses can be taken closer together to avoid night-time dosing. Parents should be advised to contact the medical team if a dose is missed or vomited.
Vitamin A derivatives
There are two derivatives of Vitamin A used as paediatric anti-cancer treatments. Cis Retinoic Acid, also called Isotretinoin, and All Trans retinoic acid, also known as tretinoin. These treatments are known as differentiating agents as they force cancer cells to mature and thus stop growing.
Both agents are available in capsules. For children who cannot swallow capsules, there is also an unlicensed medicine (ULM) suspension available. However, the lead time may be lengthy, so advanced planning is required. Alternatively, the gelatin capsules can be pierced and the liquid inside, consisting of the drug in soyabean oil, can be mixed with warm milk to create a suspension.
The hospital pharmacist will provide counselling to parents on advance ordering from their community pharmacy, formulation manipulation if required, administration and side-effects.
Mercaptopurine
Mercaptopurine, also known as 6-MP, is a thiopurine that is used in the active and maintenance treatment of ALL. It is an oral prodrug that is metabolised to active thioguanine nucleotides. These nucleotides are incorporated into DNA, compete with natural guanine, and ultimately lead to cell apoptosis.
Mercaptopurine is available as a licensed 20mg/mL liquid, licensed 50mg tablets and ULM 10mg tablets. Doses will be rounded to a measurable value depending on which formulation the patient prefers.
As the mercaptopurine 10 mg tablet is an ULM, the code does not appear on the main GMS list. For LTI patients with a confirmed Acute Leukaemia (illness code N) it is available under a triple 7 code. In this case, the community pharmacy must call the PCRS to inform them at what price they are obtaining the medicine and to get approval to use that code for the patient.
When a patient is started on mercaptopurine the hospital pharmacist will provide counselling to the patient/parents on the following:
- Safe handling;
- Mercaptopurine displays diurnal variation in pharmacokinetics and efficacy. Administration in the evening compared to morning administration may lower the risk of relapse;
- Patient must be consistent with taking with or without food. Although the SPC recommends avoiding dairy products as they may affect absorption, the patient does not need to completely avoid them, but should be consistent;
- Side effects to watch out for: hypoglycaemia, photosensitivity, diarrhoea, vomiting and nausea; and
- Advise parents to contact medical team if a dose is missed or vomited.
Cyclophosphamide
Cyclophosphamide is an alkylating agent that works by interfering with DNA replication, leading to cell death, particularly in rapidly dividing cancer cells. It is a chemotherapy medicine commonly used to treat certain cancers and leukaemia. It is available as 50 mg tablets or as an unlicensed liquid supplied from the UK, which usually has a lead time of around seven working days.
Cyclophosphamide can irritate the bladder wall, causing symptoms such as pain on passing urine, dark-coloured urine, or blood in the urine. For this reason, it should be taken in the morning with plenty of water, and good hydration should be maintained throughout the day. The dose and duration of treatment depend on the diagnosis and the chemotherapy protocol being used.
Counselling
Safe handling of oral chemotherapy agents
As many of the oral chemotherapy agents are cytotoxic in nature, thorough counselling on the safe handling of these agents is always provided to parents and carers in order to protect family members who may be in contact with the drug. The Children and Young People’s Cancer Association (CCLG) (see cclg.org.uk), provide information leaflets on safe handling of cytotoxic agents. The information in these leaflets cover:
- Storage;
- Handling precautions;
- Managing spillage on body, work surface or clothing;
- How to dispose of bodily fluids; and
- How to dispose of expired or unused medicine.
Counselling charts
On first discharge from CHI, each patient will be provided with a medication chart for all supportive care medicines. A chart will also be provided for new oral chemotherapy agents. Community pharmacists are encouraged to liaise with the hospital pharmacist if a copy of the chart is required.
Useful resources
- CHI formulary, Clinibee;
- BOPA website — formulation information, bopa.org.uk;
- CCLG website — PIL on safe handling of cytotoxic agents, cclg.org.uk; and
- Local hospital pharmacist with responsibility for haem-onc services — clarification on any prescription queries.
References available on request.
Article series on the role of the pharmacist in Paediatric Cancer Services
Part 1 of this series, a feature on
‘Paediatric Cancer Services: the role of the clinical pharmacist’ was published in the February 2026
IPU Review and is available at
ipu.ie/ipu-review.


