Home » IPU Medicines Shortages Survey 2024: Patients protected by pharmacists’ work to manage medicine shortages
The IPU has conducted an annual medicine shortage survey with its members for a number of years. This year the survey was conducted during the month of February and in this article Clare Fitzell, the IPU’s Head of Strategic Policy, discusses the survey results and what this means for Irish patients and community pharmacists supporting their care. She also discusses the ongoing work being undertaken to mitigate shortages at a national and European level and asks the important question, is enough being done at a fast enough pace to protect patients?

Hardly a week goes by without an article in the Irish media highlighting a medicine shortage, but none has gained as much attention as Ozempic. The limited supply of this drug continues to be a difficulty for patients, pharmacists and prescribers, with an inability to meet the needs of all patients prescribed this medication on a regular basis. Ask any community pharmacist, what is the most common telephone query they have on a daily basis, and they will all reply, ‘do you have Ozempic in stock’? The demand for this medicine is exceptional, the likes of which are reminiscent of the early COVID-19 pandemic where the public was searching high and low for face masks and hand sanitiser.
The root causes of medicine shortages are diverse and multi-factorial ranging from: globalisation of active pharmaceutical ingredient (API) production; shifting demands; national pricing strategies; allocations (quotas); regulatory issues; and supply distribution problems — to name but a few. Basically, you could say it’s complicated, with many inter-related factors at play with no one solution going to fix this problem. However, that doesn’t mean we shouldn’t try! The results of this year’s IPU Medicines Shortages Survey show that we may be seeing a slowdown in the downward trend progression. While the cynic in me would say we have hit rock bottom, and the only way is up, only time will tell.
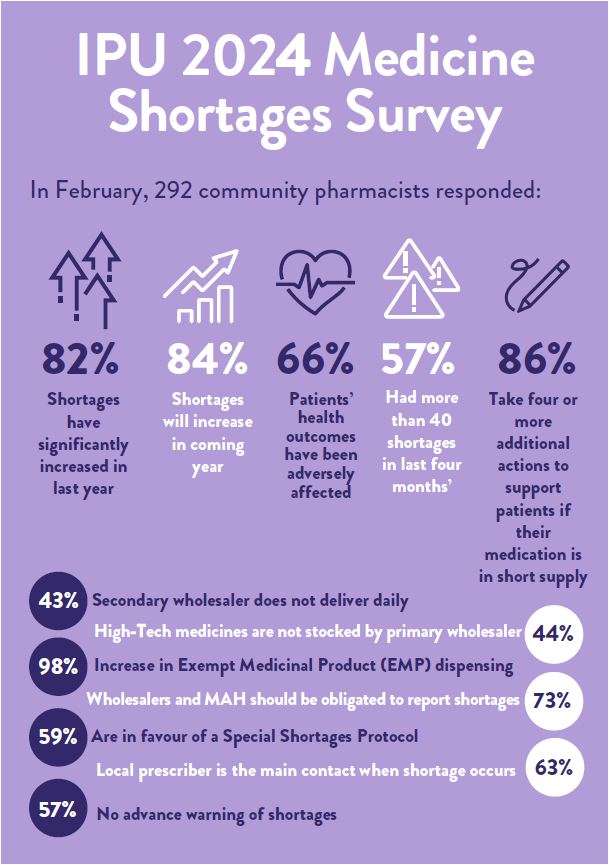
The IPU’s annual Medicine Shortages Survey was conducted in February and 292 responses were received, which is in-line with responses to last year’s survey. In this year’s survey we asked additional questions, which have provided insights on how pharmacists are managing shortages in practice, and their preferred communication preferences with patients and prescribers. Figure 1 indicates a very slight improvement on the number of medicine shortages being encountered by community pharmacists. This may be a reflection on the 2023 survey, which was conducted a month earlier in January 2023, when we were experiencing significant supply constraints of antibiotics, which thankfully were not noted on the Irish market this winter season. However, the reality behind every shortage is a patient, who either has to wait for a medication, wait for a pharmacist and prescriber to have a discussion on an alternative, or pay out-of- pocket for an exempt medicinal product that is not reimbursed under our community drug schemes. Each medication switch as a result of a shortage for a patient requires additional counselling requirements and information to be passed to the patient, to ensure the safe and effective use of the medicine, and so that patient is aware that the white capsule is now a pink tablet etc, etc.
Figure 1: Approximately, how many shortages have you encountered in the last four months?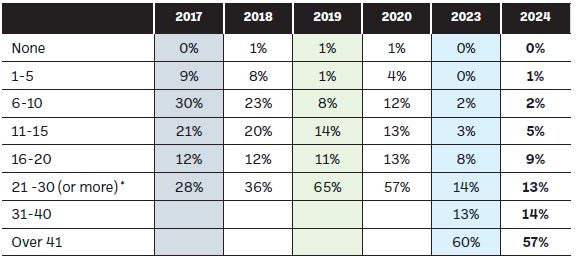 (*please note additional fields were added in 2023 due to the large volume in the upper category in previous years’ survey, i.e. 2017-2020)
(*please note additional fields were added in 2023 due to the large volume in the upper category in previous years’ survey, i.e. 2017-2020)
When pharmacists were asked to predict the future of medicine shortages over the next 12 months, as outlined in Figure 2, they were slightly less pessimistic than previous years, although the figures predicting an increase in shortages over the next 12 months remain the overwhelming majority.
Figure 2: In your opinion, in the next 12 months, will medicine shortages: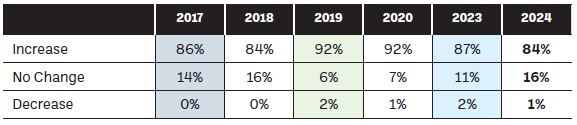
Sentiments expressed by community pharmacists as to why they predicted an increase included:
Figure 3 below indicates the main reasons community pharmacists believe to be causing medicine shortages. As indicated in the introduction, there is a wide spread of causes being indicated, with community pharmacists highlighting allocation, national pricing policies and manufacturing issues as the top reasons for shortages to be occurring.
Figure 3: What, in your opinion, are the main causes of medicine shortages? (you can indicate more than one answer)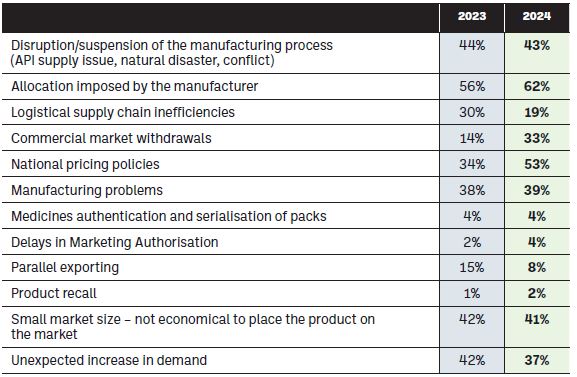
Although allocation was cited as the top reason for short supply, 82 per cent of pharmacists surveyed believed this to be an appropriate way to manage a medicine in short supply, which was an increase from the 2023 figure, where 77 per cent indicated it was appropriate.
81 per cent of community pharmacists indicated that they struggled to receive medicines from wholesalers in a timely manner, even when they were not out-of-stock at a national level, with many indicating that they were now having to check wholesaler websites multiple times daily to access medications. When asked for the reasons for this, an overwhelming majority of 93 per cent indicated that they were struggling due to the medicine being on allocation, and the next highest reason was that High-Tech Medicines were not stocked by their primary wholesaler (44 per cent).
The average time spent on a monthly basis is consistently increasing year-on-year, as reflected in Figure 4, with 25 per cent of community pharmacists now indicating that they are spending over 30 hours a month resolving medicine shortages. Figure 4: What is the average time spent per month by you and/or other pharmacy staff in resolving medicine shortages?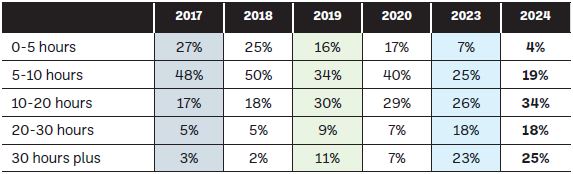
To allow better comparison to European figures we asked community pharmacists for the first time to equate the time spent on a weekly basis managing medication shortages.
Irish community pharmacists are spending 4 hours 37 minutes per week managing medicine shortages.
To get a better understanding of how pharmacists across Ireland are supporting patients when their regular medication is in short supply, we asked them to indicate the measures they take to ensure patient outcomes are not adversely impacted, and the results are revealed in Figure 5.
 Figure 5: Please indicate the measures you take to ensure patient outcomes are not adversely impacted when a medicine is in short supply?
Figure 5: Please indicate the measures you take to ensure patient outcomes are not adversely impacted when a medicine is in short supply?
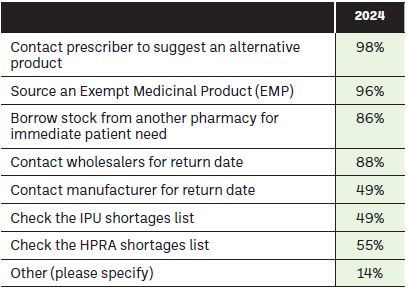
The commitment by community pharmacists to managing this issue is clear, with over 86 per cent of respondents indicating that when faced with a drug shortage issue, they use four or more different approaches to minimise the impact of this on their patients. It must be mentioned that these actions go largely unseen by patients and the additional associated workload is not recompensed. In fact, the additional workload of achieving reimbursement for GMS patients through the discretionary hardship arrangements for exempt medicinal products without administrative codes is mind boggling, inefficient, and it is the luck of the draw whether or not your pharmacy is located in an area where the local hardship office is efficient, or not, in processing payments under this arrangement. Whilst we are on the subject, the fact that we even have administrative codes for exempt medicinal products suggests that we have an issue with our reimbursement system, and surely this is worthy of closer examination?
The 14 per cent who cited ‘other’ strategies that they employ to mitigate shortages, as indicated in Figure 5, included:
It is clear from my privileged position of having an overview of all the comments made in the survey by community pharmacists, that the commitment being made the length and breadth of this country by community pharmacists to support patients find alternative medicines, should be commended. Personally, I am very proud of our profession and this tireless work we, do day-in day-out, to support our communities.
We also asked an additional question this year on the preferred communication method between community pharmacists and prescribers in relation to medicines in short supply, and the results are indicated in Figure 6 below, with healthmail being the communication choice of preference.
Figure 6: If you contact a prescriber to suggest an alternative product when a medicine is in short supply, please indicate your communication preference.

The trend on the number of exempt medicine products pharmacists are dispensing continues to rise year-on-year, with the overwhelming majority of community pharmacists indicating that this has increased annually, as outlined in Figure 7. The reasons cited for this increase range from medicine shortage of originator products to products not being licensed in Ireland, to products no longer marketed. However, it is worth noting that pharmacists indicated that it is difficult for them to ascertain if a medicine is no longer available on the market, as they are given no formal notification in this regard.
“Medicines placed on the Irish market must be authorised by the HPRA or, in the case of centrally authorised products, by the European Commission. However, European regulations do provide for an exemption to this rule – this is via Article 5 of Directive 2001/83/EC. In this case, unauthorised medicines may be supplied where a registered doctor or dentist has prescribed the product for an individual patient under his or her direct responsibility. This is in order to fulfil the special needs of those patients. Such products are defined as ‘exempt medicinal products’.”
Available at hpra.ie > Medicines > Regulatory Information > Medicines Authorisation > Notification System for Exempt (unauthorised) Products.
Figure 7: Has there been an increase in the number of unlicensed medicines (exempt medicinal products) you dispense?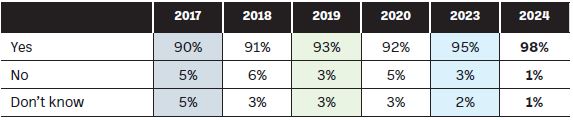
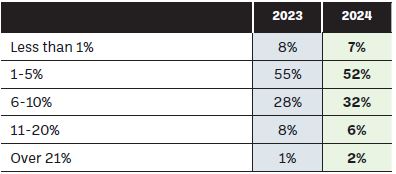
Figure 8 highlights the increased trend in the percentage of exempt medicinal products being dispensed by community pharmacists. Given the extra record keeping, legislative and administration requirements for these medicines, the additional workload should not be underestimated. A question that crosses my mind, is who is tracking the amount of these medicines now being dispensed to patients across Ireland, and are we factoring the cost of reimbursing these under our medicines budget? I for one have not seen the associated increased cost of funding these medicines highlighted in any national report to date.
Figure 8: What percentage of your dispensary items are unlicensed (exempt medicinal products)?
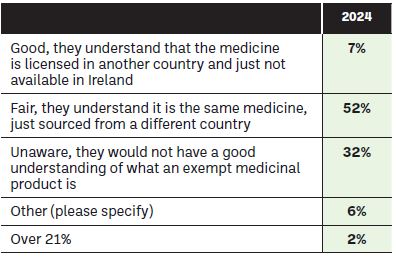
As for patient awareness, we have some collective work to undertake along with our stakeholders on patients’ understanding of why they are receiving an exempt medicinal product. Figure 9 highlights that the majority of community pharmacists indicated that patients did not have a good understanding of what an exempt medicinal product is.
Figure 9: What in your opinion is a patient understanding of an exempt medicinal product (EMP), when you are counselling them on the need for the use of this product(s)?
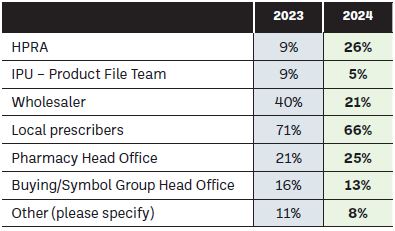
When asked do you report shortages on a regular basis, only 24 per cent of respondents indicated yes, a reduction from 32 per cent on previous years. Those pharmacists who indicated that they did report shortages indicated a variety of places they reported shortages to, as indicated in Figure 10. With much talk in European circles of medicines shortages reporting, it is clear that centralisation and automation of this process could lead to an improvement on the current disparate nature and variety of ways, which community pharmacists utilise to communicate shortages.
Figure 10: If you ticked yes to the above question, who do you report shortages to? (tick any that apply)
You can report shortages directly to our IPU Product File team at datainfo@ipu.ie, who will investigate the reason for shortage, and the anticipated return date. The IPU Medicines Shortages Report is searchable and is updated almost every day. It can be accessed ipu.ie > Members Support > IPU Product File > Medicine shortages. Additionally, you can download a form from the HPRA website and report the shortage directly to the regulator, go to hpra.ie > Medicines > Medicines Information > Medicines Shortages > Reporting a medicine shortage.
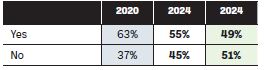
Figure 11 highlights that 49 per cent of pharmacists have signed up to receive HPRA alerts on medicine shortages, down slightly on the 2023 figure of 55 per cent.
Figure 11: Are you signed up to receive alerts from the HPRA on medicine shortages?
Community pharmacists indicated reasons for not signing up to receive alerts from the HPRA and these are highlighted in Figure 12.
Figure 12: If not, why have you not signed up for HPRA alerts?

It is worth noting that analysis of IPU website user information indicates the most visited page of our website is the medicines shortages update page, which at the time of writing this article had 411 entries. The HPRA have updated the look and feel of their medicine shortages list and added additional features, making it easier to search for information.
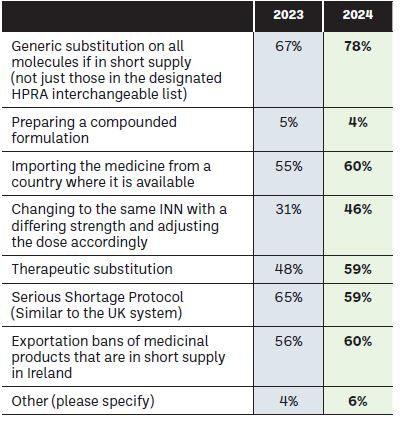
Community pharmacists indicated their support for a variety of measures that could be adopted to better support the management of medicine shortages, which are highlighted in Figure 13, with generic substitution being the preferred solution.
Figure 13: What solutions would you favour in the management of medicine shortages in Ireland? (you may indicate more than one answer)
Given the results of the survey, it would be difficult to answer this question affirmatively, however we are aware of several legislative amendments currently contained within the Health (Miscellaneous Provisions) Bill 2024 (available at oireachtas.ie > Bills & Acts), currently at second stage of the legislative process, that have some promise to improve the situation into the future. These include:
The HPRA oversees a multi-stakeholder framework, first established in 2018, which is intended to address the issue of human medicine shortages in Ireland. The aim of the framework is to:
The framework sets out the roles of different stakeholder groups in managing shortages. Participants in the framework include:
The framework outlines procedures for notification of shortages, their evaluation and communication. It also looks to further develop strategies that can help prevent shortages. The HPRA conducted a two-year review of the medicine shortages framework in November 2022, and as a result of this review made a series of recommendations, and some of the legislative updates above are a result of these recommendations. The IPU is supportive of the HPRA multi-stakeholder approach and we work constructively with our colleagues across the supply chain to advocate for transparency and equitable supply for patients across Ireland.
Medicine shortages are of an equally significant level of concern at a European level as they are at a national level in Ireland, with most European countries reporting significant constraints on their medicine supply chains.
In December 2023, PGEU (the Pharmaceutical Group of the European Union, of which the IPU is a member), conducted a survey of members to understand the worsening situation across Europe. The results of the survey were published early in 2024; the most significant statistic from this report is that each pharmacy across Europe now spends on average 9.5 hours per week managing shortages — this is a considerable increase on the 2022 figure, which was 6.7 hours.
In October 2023, the European Commission adopted a Communication on medicines shortages. This communication outlines a number of initiatives across the EU to help mitigate the problem of medicine shortages, some of which are listed below:
The examples are too numerous to mention. What is clear on reading the comments by community pharmacists is that behind all the facts and figures mentioned in this article are patients. Patients who are unable to obtain medication to treat and manage their health optimally and supporting them is a community pharmacist working tirelessly to minimise the impact on their health and prevent adverse outcomes.
Provided by Áine McCabe, McCabes Pharmacy
Rasagiline is an irreversible inhibitor of monoamine oxidase used for the symptomatic management of Parkinson’s disease as monotherapy, or as adjunct therapy with levodopa. For many patients with Parkinson’s disease, who are stabilised on this drug, rasagiline helps them to maintain mobility, independence and a good quality of life.
Since late summer 2022 there have been ongoing shortages reported for this medicine. At one point in late 2023 there was no generic, PI or exempt medicinal product (EMP) version available at all. This lasted for approximately five weeks.
A pharmacist within the McCabes pharmacy group had five patients who were prescribed this medicine. Due to the ongoing issues with supply the pharmacist contacted them all individually to assess what supply they had, so as to allow the pharmacist to prioritise the stock they had left.
All patients were extremely worried about the lack of supply and how they would cope without it. They could not comprehend how the ‘Government’ could allow this important medicine to be unavailable to them, and how there was ‘no’ supply to be had.
The pharmacist took the time to explain all the possible steps they take to try and source the medicine for them, including buying from a UK company, trying PI suppliers in Ireland, asking other pharmacies in the area, and ringing the manufacturer directly. However, they also had to flag that, it was becoming more and more common, that in spite of all the steps described above, the medicine could not be sourced.
The pharmacist then moved to dispensing weekly supplies to their patients, and also informed the prescribers so that they would have time to consider alternatives to rasagiline in case the pharmacist couldn’t get more supplies in time.
Switching products for patients stabilised on this medicine is a difficult and time-consuming process. There could be patient safety risks around fluctuations in their condition for patients, which may limit their mobility or put patients at increased risk of falls.
An EMP version became available from one company, but it was much more expensive than those with reimbursement codes. When the PCRS were contacted to ask if they would reimburse it, they refused, so for patients who did not have the means of additional payment, this was not a viable alternative.
The pharmacist then ran out of all stock of rasagiline. However, about four weeks after this, a generic became available, and the pharmacist was able to fill the prescriptions. Two of the patients were not happy to have to change brand, but understood it was that or nothing. It caused them a great deal of anxiety and the pharmacist was asked weekly by these patients if the brand had come back in.
Then after about three months, this product went short, resulting in another switch to an exempt medicinal product version. This created confusion and more anxiety for these patients.
This situation is still ongoing now five months later. Unfortunately, these situations are now not unique and happening across many medications, with significant impact on patients, and significant impact on pharmacists and prescribers supporting their patients.
Clare Fitzell

Head of Strategic Policy, IPU
Highlighted Articles